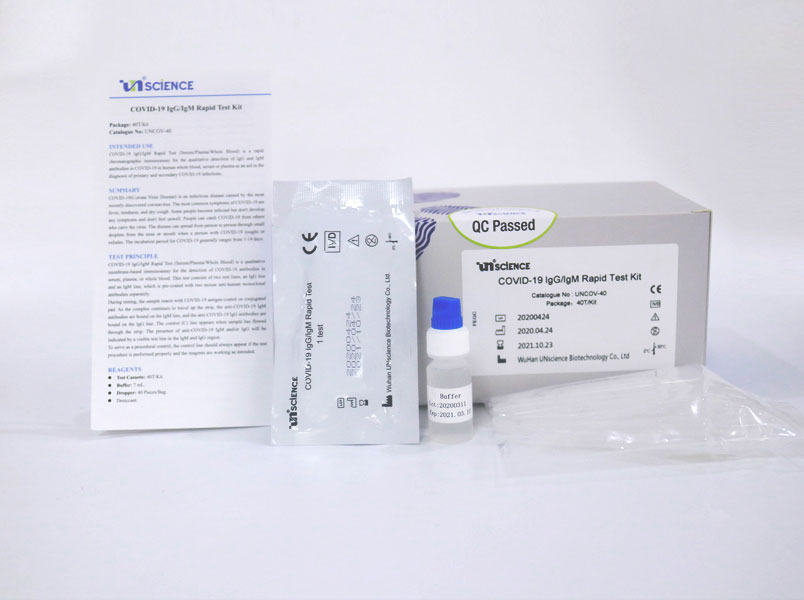
COVID-19 IgG/IgM Rapid Test Kit
- Cat.No.:UNCOV
- Sample Type:Serum/ Plasma/ Whole blood
- Lead Time:Order now, ship in 3 days
Product Introduction
COVID-19 IgG/IgM Rapid Test (Serum/Plasma/Whole Blood) is a qualitative membrane-based immunoassay for the detection of COVID-19 antibodies in serum, plasma, or whole blood. This test consists of two test lines, an IgG line and an IgM line, which is pre-coated with two mouse anti-human monoclonal antibodies separately. During testing, the sample reacts with COVID-19 antigen-coated on conjugated pad. As the complex continues to travel up the strip, the anti-COVID-19 IgM antibodies are bound on the IgM line, and the anti-COVID-19 IgG antibodies are bound on the IgG line. The control(C)line appears when sample has flowed through the strip. The presence of anti-COVID-19 IgM and/or IgG will be indicated by a visible test line in the IgM and IgG region. To serve as a procedural control, the control line should always appear if the test procedure is performed properly and the reagents are working as intended.
Kit Components & Storage
Storage
The kit can be stored at 2-30℃ for 18 months. Do not use after the expiration date.